
Platelet-rich plasma therapy—PRP—has gone from a niche procedure used in elite sports medicine to one of the most widely offered regenerative treatments in the country. You can find it at orthopedic offices, aesthetic clinics, hair restoration centers, and medical spas. The problem is that the label “PRP” on a clinic’s menu tells you almost nothing about the quality of what you are actually going to receive.
PRP is not a single standardized product. It is a category of biologic treatment, with enormous variability in preparation, composition, concentration, delivery, and clinical application. The difference between a well-prepared, precisely delivered PRP injection and a poorly prepared one injected without guidance is not a minor nuance—it is the difference between a meaningful therapeutic outcome and an expensive disappointment.
This article from the experts here at InterveneMD covers everything you need to understand about PRP: what it is, how it works biologically, where the evidence supports its use, and—critically—the questions you need to ask and the standards you need to insist on before committing to treatment. Because in the current landscape of PRP in Charleston, the quality of the clinic matters as much as the therapy itself.
PRP is one of the most promising tools in regenerative medicine. It is also one of the most inconsistently practiced. Understanding the difference between excellent and adequate PRP is the most important thing you can do before pursuing treatment.
What Is PRP, and Where Did It Come From?
Platelet-rich plasma is a concentration of platelets derived from a patient’s own blood. Platelets—the tiny cell fragments best known for their role in blood clotting—are also the body’s primary first responders to tissue injury.
When tissue is damaged, platelets flood the site and release a complex payload of growth factors and signaling proteins that initiate and orchestrate the healing cascade. These include platelet-derived growth factor (PDGF), transforming growth factor beta (TGF-β), vascular endothelial growth factor (VEGF), insulin-like growth factor (IGF-1), fibroblast growth factor (FGF), and epidermal growth factor (EGF), among others.
By concentrating platelets from a patient’s blood and delivering them directly to a site of an injury or degeneration, PRP essentially amplifies the body’s own healing signals—flooding damaged tissue with a concentrated dose of the very molecules that drive repair, collagen synthesis, angiogenesis, and cellular regeneration.
The History of PRP
The origins of PRP as a medical therapy trace back to the 1970s, when hematologists first developed methods for concentrating platelets from whole blood. Its earliest clinical applications were in oral and maxillofacial surgery—dentists and oral surgeons in the 1980s and 1990s began using PRP to accelerate bone and soft tissue healing following dental implants, extractions, and reconstructive jaw procedures. The results were compelling enough that the technology migrated into orthopedic surgery, where it was initially used to accelerate wound healing and tendon repair following procedures.
The pivotal moment that brought PRP into mainstream awareness came in the mid-2000s, when reports emerged that professional athletes—including prominent NFL and MLB players—were using PRP injections to accelerate recovery from tendon and ligament injuries. The combination of high-profile athletic use and compelling early clinical data drove rapid expansion into sports medicine, orthopedics, aesthetics, and hair restoration.

Today, PRP is one of the most commonly performed regenerative procedures globally—with applications spanning orthopedics, spine medicine, aesthetics, hair loss, sexual health, and wound care.
The Biology of Healing: How PRP Actually Works
Understanding why PRP works—and why preparation quality matters so much—requires understanding the biology of tissue repair. When injury occurs, healing unfolds in three overlapping phases: inflammation, proliferation, and remodeling. Each phase depends on specific cellular signals, and platelets are central orchestrators of all three.
In the inflammatory phase, platelets release cytokines and growth factors that recruit immune cells to the injury site, signal the need for repair, and begin breaking down damaged tissue.
In the proliferation phase, platelets stimulate fibroblasts to produce collagen, endothelial cells to form new blood vessels, and stem cells to migrate to the repair site.
In the remodeling phase, the new collagen matrix is organized and strengthened under the continued influence of platelet-derived signals.
By delivering a concentrated bolus of platelets directly to a site of chronic degeneration—a joint that is no longer healing on its own, a tendon that has become fibrotic and poorly vascularized, a scalp where follicles are miniaturizing—PRP essentially restarts a healing process that has stalled. It does not add anything foreign to the body. It amplifies and redirects the body’s own repair biology.
This autologous nature—the fact that PRP comes entirely from the patient’s own blood and goes back into the same patient—is one of its most compelling attributes. There are no donor antigens, no risk of disease transmission, no immune rejection, and no concerns about using biological material derived from another person’s cells. In an era of increasing sophistication about what we put into our bodies, the appeal of a therapy that is entirely self-derived is significant and well-founded.
Not All PRP Is the Same: The Critical Variables That Determine Outcomes
One of the most important and least discussed variables in PRP quality is the leukocyte— white blood cell—content of the final product.
Leukocyte-Rich vs. Leukocyte-Poor PRP—and Why It Matters
PRP preparations are broadly categorized as either leukocyte-rich PRP (LR-PRP) or leukocyte-poor PRP (LP-PRP), and the distinction has significant clinical implications that are actively debated in the research literature.
Leukocyte-rich PRP contains concentrated white blood cells alongside the concentrated platelets. Leukocytes—particularly neutrophils and monocytes—contribute additional growth factors and immune signaling molecules to the preparation, and they play a role in the antimicrobial defense of the injection site. However, they also contribute pro-inflammatory cytokines, including interleukin-1 beta (IL-1β) and tumor necrosis factor-alpha (TNF-α), which can amplify the inflammatory response at the injection site.
Leukocyte-poor PRP, by contrast, is processed to minimize white blood cell content, producing a preparation that is predominantly platelets in plasma with significantly reduced inflammatory signaling. The reduced pro-inflammatory load is thought to be beneficial in environments where excess inflammation is counterproductive—most notably in intra-articular (joint space) injections, where the synovial environment is sensitive to inflammatory stimulation and where excess inflammation can cause post-injection flares and potentially accelerate cartilage degradation.
The current weight of evidence generally supports LP-PRP for intra-articular applications—particularly knee osteoarthritis, where multiple studies have shown LP-PRP to be superior to LR-PRP for pain reduction and functional improvement.
For tendon and ligament injections, the picture is more nuanced, with some research suggesting LR-PRP’s more robust inflammatory signal may be advantageous in promoting the initial healing cascade in poorly vascularized structures.
The critical point is that knowing which type of PRP is appropriate for a given application— and having the ability to produce it consistently—requires both clinical knowledge and technical capability. Many clinics offering PRP in Charleston have neither the processing equipment nor the clinical expertise to reliably produce one type versus the other. They produce whatever their centrifuge and kit generate, without characterization of what that product actually contains. At InterveneMD, we can differentiate and that makes all the difference for our patient.
The Volume Problem: How Much Blood Are They Drawing?
Here is one of the most practically important questions you can ask any clinic offering PRP, and it is one most patients never think to raise: how much blood are you drawing?
The answer matters enormously. Platelets cannot be manufactured or added from an external source—they come entirely from the patient’s own blood. The only way to get more platelets into the final PRP product is to start with more blood. More blood drawn means more platelets available to concentrate. And the number of platelets in the final product is one of the primary determinants of therapeutic efficacy.
A disturbingly large number of clinics offering PRP draw approximately 15 to 30 milliliters of blood—roughly one to two tablespoons. After centrifugation, the resulting PRP volume and platelet count from such a small draw is, in the frank assessment of practitioners who take this seriously, inadequate for meaningful tissue repair. It may be enough to generate a result that feels like something happened. It is rarely enough to drive genuine regenerative outcomes in damaged musculoskeletal tissue.
Clinics serious about PRP outcomes draw significantly more blood. A minimum of 120 milliliters is a reasonable threshold for musculoskeletal applications—and many advanced protocols draw considerably more, particularly when treating large joint complexes or multiple sites.

Before committing to PRP in Charleston or anywhere, ask your provider directly: how many milliliters of blood will you draw? If the answer is under 100 mL, that is a red flag about the seriousness with which that clinic approaches the therapy.
The Cell Counter: The Gold Standard for PRP Characterization
Beyond the volume of blood drawn, the next critical quality variable is whether the clinic actually knows what is in the PRP they are injecting. This is where the vast majority of PRP providers fall short—and where the gap between adequate and excellent becomes most stark.
A cell analyzer—essentially a CBC differential counter, the same type of instrument used in hospital hematology labs—can precisely quantify the platelet count and white blood cell composition of both the original blood draw and the final PRP product. This tells the clinician exactly how many platelets the patient started with, how efficiently the centrifugation process concentrated them, what the final platelet concentration is in the product being injected, and what the leukocyte composition of that product is.
This information is not a luxury. It is the foundation of precise, reproducible, outcome-driven PRP medicine. Without it, the clinician is essentially injecting a biological product of unknown composition and hoping for the best. With it, they can verify that the product meets minimum platelet concentration thresholds for the intended application, adjust protocols based on individual patient platelet characteristics, and track product quality systematically across patients.
Here is the uncomfortable reality: fewer than 10 percent of clinics offering PRP use cell analyzers to characterize their product. The primary reason is cost—high-quality cell analyzers represent a significant capital investment that most providers are unwilling or unable to make. The secondary reason is that most PRP providers are not operating at the level of clinical rigor where this distinction matters to them. At InterveneMD however, we did make this investment when we decided to offer PRP in Charleston.
When evaluating a clinic for PRP treatment, asking whether they use a cell analyzer to count and characterize their product before injection is one of the single most revealing questions you can ask. It instantly differentiates the small minority of practices that are doing this with genuine scientific rigor from the majority that are not. The answer tells you a great deal about how seriously that clinic takes the therapy they are offering.
Most clinics do not know what is actually in the PRP they inject. They draw a small volume of blood, spin it, and inject the result without ever characterizing the product. Insisting on a clinic that uses a cell analyzer is not being overly demanding—it is the minimum standard for evidence-based care.
Delivery Matters: Why Imaging Guidance and Physician Expertise Are Non-Negotiable
The biological quality of the PRP product is only meaningful if it is delivered to the right location. This sounds obvious. In practice, it is one of the most commonly violated principles in PRP delivery.
Image Guidance: Fluoroscopy and Ultrasound
The human body is not transparent. Joint spaces, tendon sheaths, spinal facet joints, nerve root sleeves, and the specific tissue targets for most PRP injections are small, anatomically variable, and located beneath layers of muscle, fat, and connective tissue that make blind injection—injection without imaging guidance—an exercise in approximation at best. Studies using post-injection imaging to verify needle placement in blind injections have consistently found that a significant percentage of ostensibly intra-articular injections miss the target—with needles ending up in periarticular tissue, the joint capsule, or entirely outside the intended structure.
For spine injections, the stakes are even higher. The proximity of major vascular structures, nerve roots, and the spinal cord to the target injection sites means that imprecise needle placement is not just ineffective—it can be dangerous. Fluoroscopy (real-time X-ray guidance) and diagnostic ultrasound both allow the physician to visualize the needle in real time as it approaches and enters the target structure, confirming precise placement before injection. Contrast dye under fluoroscopy additionally allows visualization of the spread pattern of the injectate, confirming that the material is distributing appropriately within the target space.
There are practices performing PRP injections—including spinal PRP injections—without any imaging guidance whatsoever. This is not a defensible clinical practice. It reduces the likelihood of accurate delivery, increases the risk of adverse events, and ultimately undermines the therapeutic potential of even a high-quality PRP product. Imaging guidance is not an optional upgrade. It is a standard of care.
Physician-Performed Injections
Equally important is who is holding the needle. PRP injections—particularly for complex orthopedic and spinal applications under imaging guidance—are advanced procedural medicine. They require an understanding of anatomy, pathology, imaging interpretation, and injection technique that is built over years of medical training and clinical experience.
In the current landscape of PRP delivery, injections are being performed by practitioners across a wide spectrum of training: fellowship-trained interventional spine physicians, orthopedic surgeons, sports medicine physicians, mid-level providers including nurse practitioners and physician assistants, and in some settings, staff members with minimal procedural training. The variability in skill and knowledge across this spectrum is enormous.
A physician who has performed thousands of image-guided injections, who understands the nuances of spinal and joint anatomy, who can read the imaging in real time and adjust technique accordingly, and who has the clinical judgment to recognize when a finding on imaging changes the treatment plan—that physician is offering a fundamentally different service than a mid-level provider who learned the procedure in a weekend course and is performing it under limited supervision.
For elective, cash-pay procedures like PRP—where you are investing meaningful money and placing significant hope in the outcome—the training and experience of the injecting physician is not a trivial consideration. It may be the most important variable of all. Seek out the most experienced, most highly trained physician available for these procedures. Ask about their training, their volume of procedures, and their use of imaging guidance. The answers will tell you a great deal about what kind of outcome to expect.
Preparing the Body: The Step Most Clinics Skip Entirely
Here is a principle that is fundamental to regenerative medicine and almost universally ignored in the way PRP is delivered in most clinical settings: the tissue being injected lives in a body. And the biological environment of that body— the health of the immune system, the function of the mitochondria, the balance of the hormonal system, the state of the gut microbiome—profoundly influences whether the healing signal delivered by PRP can actually be acted upon.
PRP works by delivering growth factors and signaling molecules that instruct cells to repair damaged tissue. But cells that are metabolically compromised, mitochondrially depleted, hormonally disrupted, or operating under chronic inflammatory burden from gut dysbiosis do not respond to repair signals with the same efficiency as cells that are healthy, well-fueled, and operating in an optimized biological environment. The injection may deliver the signal perfectly. If the cells receiving that signal are not capable of executing on it, the outcome is compromised.
Consider the analogy of planting seeds. The quality of the seed matters. But so does the quality of the soil. A high-quality PRP preparation injected into a body operating under chronic immune dysregulation, poor mitochondrial function, and hormonal deficiency is like planting excellent seeds in depleted, nutrient-poor soil. The potential is there. The outcome may not be.

A truly comprehensive approach to PRP—and to regenerative orthopedics broadly—evaluates and optimizes the patient’s systemic biological environment before the procedure.
This means:
- Assessing immune function and addressing sources of chronic immune dysregulation
- Evaluating mitochondrial health and supporting cellular energy production with targeted nutritional and supplemental protocols
- Assessing gut health, since gut-driven systemic inflammation directly impairs tissue healing capacity and cellular responsiveness
- Optimizing hormonal status, particularly testosterone and thyroid hormones, which are among the most powerful regulators of tissue repair and muscle preservation
- Correcting nutritional deficiencies—particularly vitamin D, zinc, and protein adequacy—that are directly required for the healing process
The parallel to surgical preparation is exact and intentional. Nobody would argue that you should undergo elective surgery without preparing your body appropriately—ensuring your cardiovascular health is optimized, your nutrition is adequate, your immune system is functioning, and your hormones are balanced. Yet the same patients who would insist on pre-surgical optimization routinely receive PRP injections with zero evaluation of their systemic biological readiness for healing.
If you are investing in elective, cash-pay regenerative procedures, you should be investing with the same due diligence and vigilance you would bring to surgical preparation. The procedures may be non-surgical, but the biology of healing is identical. Prepare the soil before you plant the seeds.
Peptides as a Complement to PRP
Therapeutic peptides are increasingly recognized as a powerful complement to PRP in regenerative medicine protocols—both in preparing the body for the procedure and in amplifying recovery afterward.
- BPC-157, with its documented effects on tissue repair, angiogenesis, and gut healing, directly supports the biological environment in which PRP exerts its effects.
- GHK-Cu stimulates collagen synthesis and wound remodeling through mechanisms that are synergistic with PRP’s growth factor payload.
- CJC-1295 and Ipamorelin optimize growth hormone release, supporting the anabolic environment needed for tissue regeneration.
- TB-4 promotes stem cell migration and tissue flexibility at the repair site.
Used as a pre-procedure preparation protocol and continued through the post-injection healing period, peptides address the cellular and systemic biological environment that determines how effectively the PRP signal can be executed. The combination of systemic biological optimization through functional medicine, targeted peptide protocols, and high-quality PRP delivery represents the current state of the art in regenerative orthopedic care.
The Functional Unit Principle: Why Treating One Spot Is Usually Not Enough
One of the most common limitations of PRP delivery in standard orthopedic settings is a conceptual one: the assumption that pain in an aging patient is coming from a single, identifiable, isolated source that can be targeted with a single injection.
This assumption is almost always wrong—and acting on it consistently produces suboptimal outcomes that are then incorrectly attributed to the limitations of PRP itself.
The aging musculoskeletal system does not degenerate in isolated spots. It degenerates as a system. The spine, for example, is not a collection of independent segments—it is a functional unit in which every level influences the levels above and below it, in which joint degeneration at one level creates compensatory stress at adjacent levels, in which muscle imbalances create asymmetric loading that accelerates degeneration across multiple structures simultaneously, and in which the pain experienced by the patient reflects the cumulative dysfunction of the entire system, not the pathology of a single MRI finding.
When a patient presents with chronic low back pain, the temptation—particularly in a busy clinical setting—is to identify the most impressive finding on imaging and inject it. One level. One structure. One injection. The problem is that we are not capable, with the tools currently available, of confidently attributing a patient’s entire pain experience to one isolated anatomical finding. The pain is distributed across a functional unit. Treating one small part of it while leaving adjacent degenerated structures untreated is, at best, an incomplete intervention.
Comprehensive regenerative orthopedic care treats the entire functional unit. For a patient with multilevel spinal degeneration, this may mean treating 50 or 60 injection sites across the facet joints, ligaments, tendons, and paraspinal structures of the affected region—covering the full anatomical territory that is contributing to the patient’s dysfunction. This is a fundamentally different undertaking than a single-level injection, requiring more time, more procedural expertise, greater blood draw volume to produce adequate PRP for the full treatment area, and more sophisticated clinical judgment about how to map the treatment to the patient’s specific anatomy and pathology.
The same principle applies beyond the spine. Knee degeneration involves not just the joint space but the surrounding ligaments, tendons, and muscle attachment points that contribute to joint stability and loading mechanics. Hip pain involves the joint, the labrum, the surrounding bursa, and the tendons that cross the joint. Comprehensive treatment of the functional unit—rather than point injection of a single structure—is consistently associated with better outcomes in the regenerative orthopedic literature.
An aging spine does not fail in one place. An aging knee does not fail in one structure. Treating a single spot while leaving the surrounding functional unit untreated is like replacing one worn tire on a car that needs four. The problem is the system, not the spot.
Clinical Applications: Where PRP Is Used and What the Evidence Shows
The orthopedic evidence base for PRP is the most mature and extensive of any application area.
Orthopedic and Spine Applications
Knee osteoarthritis has been studied most thoroughly, with multiple meta-analyses and systematic reviews documenting PRP’s superiority over hyaluronic acid and corticosteroid injections for pain reduction and functional improvement—with benefits that are more durable than either comparator at 12-month follow-up. Importantly, the evidence consistently favors LP-PRP over LR-PRP for intra-articular knee applications, and higher platelet concentrations are associated with better outcomes—reinforcing the importance of both product characterization and adequate blood draw volume.
Tendon pathology represents another strong evidence base for PRP. Lateral epicondylitis (tennis elbow), patellar tendinopathy, Achilles tendinopathy, and rotator cuff pathology have all been studied in randomized controlled trials, with PRP consistently showing benefits over placebo and comparable or superior outcomes to corticosteroid injections—with the important distinction that PRP’s benefits tend to improve over time as tissue repair progresses, while corticosteroid benefits are front-loaded and followed by potential tissue degradation with repeated use.
In the spine, PRP is used for facet joint arthrosis, sacroiliac joint dysfunction, discogenic pain, and ligamentous instability—often as part of comprehensive prolotherapy or prolozone protocols that treat the entire spinal functional unit. The evidence base for spinal PRP is less mature than for peripheral joint applications, but growing, and clinical experience in skilled hands consistently demonstrates meaningful outcomes for patients who have failed conventional spinal interventions.
Aesthetics: Vampire Facials, PRP Facials, and Microneedling
PRP has found significant application in aesthetic medicine, where its ability to stimulate collagen synthesis, improve skin texture, and accelerate cellular renewal has made it a popular tool for facial rejuvenation. The so-called vampire facial—made famous by celebrity endorsements in the early 2010s—involves drawing blood, processing it to PRP, and either injecting it subdermally into the face or applying it topically in conjunction with microneedling.
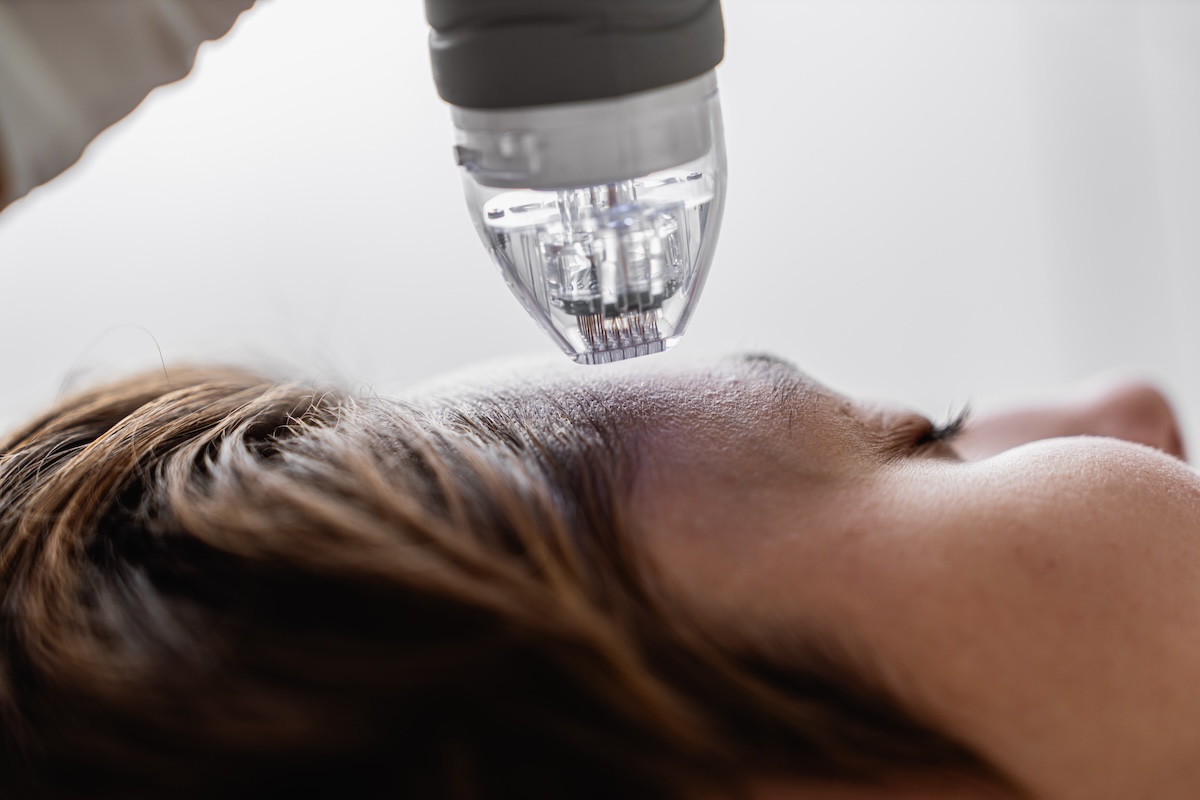
Microneedling with PRP is among the most evidence-supported aesthetic applications. Microneedling creates thousands of micro-injuries in the skin surface, triggering the wound healing response and increasing skin permeability. Applying PRP to the skin immediately after microneedling allows the growth factors to penetrate through the micro-channels and reach the dermal layer, where they stimulate fibroblasts to produce new collagen and elastin.
Multiple clinical studies have documented improvements in skin texture, fine lines, pore size, and overall skin quality with combined microneedling and PRP versus microneedling alone.
PRP is also used as a standalone facial injection—delivered subdermally across the face to improve overall skin quality, hydration, and radiance—and as an adjunct to other aesthetic procedures including laser treatments and filler injections, where it accelerates post-procedure healing and enhances outcomes.
Hair Restoration
PRP for androgenetic alopecia—the most common form of hair loss in both men and women—has emerged as one of the most promising non-surgical hair restoration tools available. The mechanism involves PRP’s growth factors—particularly PDGF, VEGF, and IGF-1—stimulating the proliferation of dermal papilla cells, improving scalp vascularity, prolonging the anagen (growth) phase of the hair cycle, and reducing the inflammation that drives follicular miniaturization in androgenetic alopecia.
Multiple randomized controlled trials and meta-analyses have documented statistically significant improvements in hair density, hair shaft caliber, and hair count following PRP treatment for androgenetic alopecia. As with musculoskeletal PRP, outcomes are highly dependent on product quality—platelet concentration, treatment frequency, and the systemic hormonal and nutritional environment of the patient all influence results significantly.
Outcome Tracking and National Registries: The Mark of a Serious Practice
One final quality indicator that separates excellent PRP practices from adequate ones is participation in national outcome registries—structured databases that track patient outcomes systematically over time following regenerative procedures. Major registries such as those maintained through the Interventional Orthopedics Foundation collect standardized outcome data from participating practices, tracking pain scores, functional outcomes, and patient satisfaction at defined intervals following treatment—typically at 3, 6, 12, and 24 months post-procedure.
Participation in these registries requires that a practice commit to the discipline of systematic outcome tracking—following up with patients, collecting standardized data, and contributing to the growing body of real-world evidence on regenerative orthopedic outcomes. Practices that participate can access their own outcome data, benchmark their results against national norms, and share that data with prospective patients considering treatment.
This matters for patients in a direct and practical way. When you consult with a practice that participates in national outcome registries, you can ask to see their outcome data—their actual results, tracked systematically over time, for patients with conditions similar to yours. That is a level of transparency and accountability that most medical practices, conventional or regenerative, do not provide. It transforms the conversation from “trust me, this works” to “here is what our data shows for patients like you.”
Ask any clinic you are considering for PRP in Charleston whether they participate in any national outcome registries. The answer, again, will tell you a great deal about how seriously they take the care they are delivering.
What to Ask Before Your PRP Procedure: A Practical Checklist
Given everything covered in this article, here are the questions every patient should ask before committing to PRP treatment anywhere:
How many milliliters of blood will you draw? The answer should be at minimum 120 mL for musculoskeletal applications. Anything significantly less raises serious questions about whether the platelet yield will be adequate.
Do you use a cell analyzer to characterize the PRP before injection? This single question separates the small minority of practices doing this with genuine scientific rigor from the majority that are not.
Will you tell me the platelet count in my baseline blood and in the final PRP product? A practice using a cell analyzer should be able to provide this number.
Is this LP-PRP or LR-PRP, and how do you decide which to use for my condition? A clinician who cannot answer this question is not operating at the level of knowledge the therapy deserves.
Will the injection be performed under imaging guidance—ultrasound or fluoroscopy? The answer must be yes for any musculoskeletal or spinal application.
Who will be performing the injection? For complex orthopedic and spinal injections, insist on a physician with specific training and high procedural volume.
Will you assess and address my systemic biological health before the procedure? A practice that treats PRP as an isolated injection without evaluating the patient’s systemic health is missing a critical component of optimal outcomes.
Do you treat the entire functional unit, or a single injection site? For spinal and complex joint degeneration, ask explicitly about their philosophy on treating adjacent and connected structures.
Do you participate in any national outcome registries, and can I see your outcomes data? The answer tells you whether the practice holds itself accountable to systematic evidence.
When done right, PRP is one of the most powerful non-surgical regenerative tools available. Poorly administered PRP is an expensive injection of unknown biological material into an unverified location. The questions above will tell you which one you are being offered.
The Bottom Line
Platelet-rich plasma therapy represents a genuine advance in regenerative medicine—a biologically rational, autologous, mechanism-driven approach to tissue repair that the research increasingly validates across a growing range of applications. From knee osteoarthritis and tendon injuries to facial rejuvenation and hair restoration, PRP’s ability to amplify the body’s own healing biology makes it one of the most versatile tools in modern non-surgical medicine.
But the gap between excellent PRP and mediocre PRP is vast—and that gap is invisible to most patients, who see only the label on the menu and assume the procedure is standardized. It is not. The volume of blood drawn, the characterization of the final product, the expertise of the injecting physician, the use of imaging guidance, the assessment and optimization of the patient’s systemic biology, the comprehensiveness of the treatment approach, and the accountability structures in place for tracking outcomes—all of these variables combine to determine whether a patient receives a genuinely transformative, regenerative intervention or an underwhelming injection that leaves them questioning whether PRP works at all.
It does work. When it is done right, by the right people, with the right preparation, in the right biological environment. That is the standard worth insisting on—and now you know the questions to ask.

Interested in PRP in Charleston? Visit the Experts at InterveneMD.
Learn whether PRP may help support healing, reduce discomfort, and improve recovery with a personalized consultation.
Key References
Dhurat R, Sukesh M. Principles and Methods of Preparation of Platelet-Rich Plasma: A Review and Author’s Perspective. Journal of Cutaneous and Aesthetic Surgery. 2014;7(4):189-197.
Mishra AK, et al. Sports Medicine Applications of Platelet Rich Plasma. Current Pharmaceutical Biotechnology. 2012;13(7):1185-1195.
Filardo G, et al. Platelet-rich plasma vs hyaluronic acid to treat knee degenerative pathology. British Medical Journal Open Sport & Exercise Medicine. 2015.
Riboh JC, et al. Effect of Leukocyte Concentration on the Efficacy of Platelet-Rich Plasma in the Treatment of Knee Osteoarthritis. American Journal of Sports Medicine. 2016;44(3):792-800.
Patel S, et al. Treatment with platelet-rich plasma is more effective than placebo for knee osteoarthritis. American Journal of Sports Medicine. 2013;41(2):356-364.
Fitzpatrick J, et al. Analysis of platelet-rich plasma extraction: variations in platelet and blood components between 4 common commercial kits. Orthopaedic Journal of Sports Medicine. 2017;5(1).
Sclafani AP, Azzi J. Platelet Preparations for Use in Facial Rejuvenation and Wound Healing. Facial Plastic Surgery. 2015;31(1):8-15.
Gentile P, et al. The Effect of Platelet-Rich Plasma in Hair Regrowth: A Randomized Placebo-Controlled Trial. Stem Cells Translational Medicine. 2015;4(11):1317-1323.
Lana JF, et al. Platelet-Rich Plasma Power: A Current Perspective. Archives of Orthopaedic and Trauma Surgery. 2021.
Andia I, Maffulli N. Platelet-rich plasma for managing pain and inflammation in osteoarthritis. Nature Reviews Rheumatology. 2013;9(12):721-730.
Kon E, et al. Platelet-rich plasma: intra-articular knee injections produced favorable results on degenerative cartilage lesions. Knee Surgery, Sports Traumatology, Arthroscopy. 2010;18(4):472-479.
Interventional Orthopedics Foundation. Registry Outcomes Data: Platelet-Rich Plasma in Musculoskeletal Medicine. IOF Annual Report. 2023.
Note:
This content is provided for educational purposes only and reflects current research and clinical thinking in functional and integrative medicine. It is not a substitute for personalized medical advice. Every person’s biology is different—what works for one individual may not be appropriate for another. If you are curious about any of the topics or therapies discussed here, we encourage you to schedule a consultation with a qualified provider who can evaluate your specific needs and goals.


